8 Minutes
Laboratory breakthrough: meteorite diamond (lonsdaleite) synthesized at scale
Scientists have for the first time produced a discernible quantity of meteorite diamond — lonsdaleite, also called hexagonal diamond — in a controlled laboratory experiment. The material, predicted by theory to be substantially harder than ordinary cubic diamond, was created as small ultrahard disks using high-pressure, high-temperature synthesis. The work, reported in Nature on July 30, 2025, marks a critical step toward validating decades of theoretical predictions about hexagonal carbon lattices and exploring their industrial applications.
Scientific background: what distinguishes lonsdaleite from ordinary diamond
Diamond is the best-known naturally occurring ultrahard material because each carbon atom forms four equivalent sp3 bonds, producing a network of tetrahedra. In the conventional cubic (face-centered cubic) diamond lattice these tetrahedra stack in three distinct repeating layers, labeled A, B and C. This three-layer ABC stacking gives cubic diamond its characteristic mechanical and electronic properties.
In contrast, lonsdaleite adopts a hexagonal crystal symmetry arising from only two repeating layers, labeled A and B, stacked in AB sequence. The difference is subtle: some carbon–carbon bond lengths become slightly shorter and others slightly longer than in cubic diamond. Crystallographers predicted that this hexagonal arrangement should increase stiffness and hardness; theoretical models have suggested a hardness increase in the range of roughly 50–60% relative to cubic diamond. Early small, impure grains tentatively identified as lonsdaleite were reported in fragments of the Canyon Diablo meteorite in the 1960s, but contamination by graphite, cubic diamond and amorphous carbon left the existence of pure hexagonal diamond in doubt for decades.
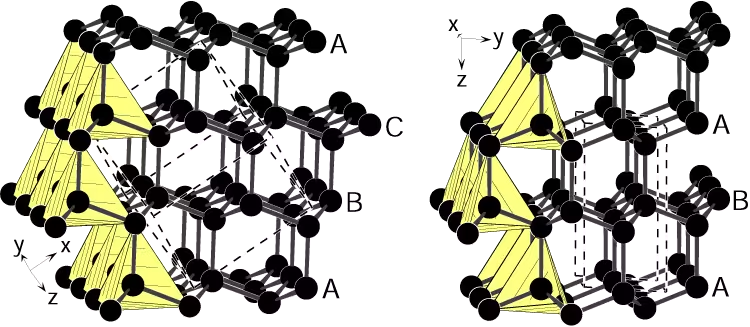
A diagram showing the structural differences between cubic diamond (left) and meteorite diamond (right). (Image credit: Ralf Riedel)
Experiment details: how researchers reproduced an impact environment in the lab
Inspired by the lonsdaleite-like grains found in the Canyon Diablo meteorite, Wenge Yang and colleagues at the Center for High Pressure Science and Technology Advanced Research (HPSTAR) in Beijing designed an experiment to mimic the extreme pressures and temperatures of meteorite impacts. The team started with high-purity graphite as the carbon feedstock and used a diamond anvil cell (DAC) to impose static pressures on the order of tens of gigapascals.
Synthesis parameters and protocol
- Pressure: approximately 20 GPa (about 200,000 atmospheres), sufficient to force adjacent carbon layers to slide and rebond.
- Heating: a tightly focused laser provided localized heating above 1400 °C to activate atomic rearrangement without causing melting.
- Kinetics: slow, controlled compression and careful, gradual decompression to lock the hexagonal stacking pattern in place and avoid back-transformation to graphite.
"At pressures around 20 GPa (200,000 atmospheres), the flat carbon layers of graphite are forced to slide and bond with adjacent layers, forming a buckled carbon honeycomb characteristic of hexagonal diamond," Yang said in communication with the press. "Laser heating above 1400 °C facilitates this transition." The laser heating and slow pressure release were critical: rapid release or inadequate thermal control risks reverting the material to graphite or generating mixed-phase carbon.
The research was inspired by a fragment of Canyon Diablo meteorite that contained lonsdaleite, which likely formed due to the high pressures and temperatures experienced during Earth impact. (Image credit: By Geoffrey Notkin, Aerolite Meteorites of TucsonOriginal uploader was Geoking42 at en.wikipedia - Transferred from en.wikipedia(Original text : Self-made. Image created by Geoffrey Notkin, Aerolite Meteorites [1]), CC BY-SA 2.5, Link)
Characterization and primary findings
After synthesis, the team applied high-resolution electron microscopy, X-ray crystallography and diffraction analysis to probe atomic structure. Transmission electron microscope (TEM) images revealed AB-layer stacking consistent with hexagonal symmetry. X-ray crystallographic patterns matched the expected hexagonal lattice of lonsdaleite, confirming that the disks included genuine hexagonal diamond domains.
Although the disks were small and not free of cubic diamond inclusions, the observations provide clear structural evidence that hexagonal diamond can be produced in the laboratory under well-controlled conditions. Hardness tests require larger, defect-free samples than those produced in this first demonstration, so the authors did not report a definitive hardness value for their samples. They did, however, show the new material is at least as hard as ordinary diamond in the limited evaluations available.
"It's a good first demonstration," said Soumen Mandal, a physicist at the University of Cardiff who studies diamond applications, in an independent comment. "Now we need pure crystals and more material to start exploring its physical and mechanical properties, thermal properties, electric properties — all of these."
Implications for industry and research
If larger, high-purity lonsdaleite crystals can be produced reliably, the material has the potential to outperform cubic diamond in several technological areas because of its predicted higher hardness and possibly distinct thermal and electronic properties. Key potential applications include:
- Industrial abrasives and drill bits for mining and oil-and-gas operations, where increased hardness directly translates to longer tool life and better performance.
- Precision machining and cutting tools that require extreme wear resistance.
- High-power, wide-bandgap electronics and thermal management systems that leverage diamond's exceptional thermal conductivity; hexagonal diamond may offer complementary electronic band structure advantages.
- Quantum technologies and sensors: diamond already hosts nitrogen-vacancy centers and other defect-based quantum systems; novel lattice symmetries could open fresh opportunities for engineered quantum defects.
The HPSTAR team estimates that practical industrial adoption could still be a decade away, given the need to scale sample size, reduce impurities, and characterize mechanical, thermal and electrical behavior comprehensively.
Expert Insight
Dr. Elena Park, a materials scientist (fictional), comments: "This is an important experimental milestone. The challenge now is scale and purity. Producing isolated AB stacking across millimeter-scale domains without cubic contamination will require refinements in pressure pathways, heating profiles and starting material control. If these obstacles can be overcome, the engineering payoff could be significant for cutting tools and thermal devices."
Dr. Marcus Olufemi, a fictional applied physicist, adds: "We should also study defect chemistry in hexagonal diamond. Defects determine not only mechanical strength but also electronic and optical properties. A new lattice symmetry means new defect behaviors — which can be tailored for specific applications in photonics or quantum sensing."
Next steps for research
To move from laboratory proof-of-principle to material platform, researchers and industry partners will need to address several priorities:
- Scale production: develop methods to synthesize larger, continuous lonsdaleite crystals, whether via static compression, shock techniques, or novel chemical pathways.
- Purity control: eliminate residual cubic diamond, graphite and amorphous carbon to enable reliable mechanical testing and device fabrication.
- Comprehensive property mapping: measure hardness (Vickers, Knoop, nanoindentation), fracture toughness, thermal conductivity, electronic bandgap, and defect behavior across a range of sample qualities and orientations.
- Economic and environmental assessment: evaluate whether the energy and equipment costs of producing hexagonal diamond at scale are justifiable for target applications.
Conclusion
The laboratory synthesis of lonsdaleite disks represents a major experimental validation of a material scientists have long sought to isolate and study. By reproducing impact-like pressures and temperatures with a diamond anvil cell and targeted laser heating, researchers created hexagonal-stacked carbon domains and verified their structure with electron microscopy and X-ray crystallography. While samples remain small and partly impure, the results reopen the path to testing a material theoretically predicted to be up to 50–60% harder than cubic diamond. Achieving larger, purer crystals is the clear next challenge; if successful, hexagonal diamond could reshape sectors from industrial manufacturing to electronics and quantum technologies over the coming decade.
Source: livescience


Leave a Comment