6 Minutes
Researchers at the University of California, Irvine report that a natural antioxidant in green tea, when combined with a common form of vitamin B3, can restore a key cellular energy molecule in neurons and stimulate the brain’s waste-removal machinery. In lab-grown mouse neurons this combination increased guanosine triphosphate (GTP) levels, revived autophagy-related cleanup functions, and reduced amyloid beta aggregates—hallmarks associated with Alzheimer’s disease. Because both compounds are available in food and as cleared dietary supplements, the findings point to a plausible, low-risk route for further therapeutic development.
Scientific background: EGCG, nicotinamide and cellular energy
Epigallocatechin gallate (EGCG) is an abundant polyphenol in green tea credited with antioxidant and neuroprotective properties. Nicotinamide is a form of vitamin B3 (a metabolite of niacin) found in cereals, fish, nuts, legumes and eggs and generated by normal cellular metabolism. The UC Irvine team investigated these two molecules together to test whether they could influence intracellular nucleotide pools—specifically guanosine triphosphate (GTP), a less-studied energy carrier that supports essential neuronal processes.
GTP participates in numerous cellular functions including microtubule dynamics, intracellular transport and autophagy—the cell’s system for degrading and recycling damaged proteins and organelles. Prior research has linked declining neuronal energy states and disrupted proteostasis with aging and neurodegenerative disease. The new experiments identify GTP decline as a potential mechanistic contributor to age-related accumulation of toxic protein aggregates.
Methods and key experimental findings
In vitro experiments used cultured mouse cortical neurons exposed to EGCG and nicotinamide, individually and in combination. The investigators measured intracellular GTP levels, markers of autophagy, and the presence of amyloid beta aggregates. They also assessed neuronal markers of age-related damage.
Combined treatment produced a measurable increase in GTP compared with controls, and neurons with higher GTP showed enhanced autophagic flux—evidence that the recycling machinery was operating more effectively. Importantly, treated cells showed reduced amyloid beta accumulation and partial reversal of cell-aging markers. These results suggest that restoring nucleotide energy balance can re-enable neurons to clear toxic proteins.
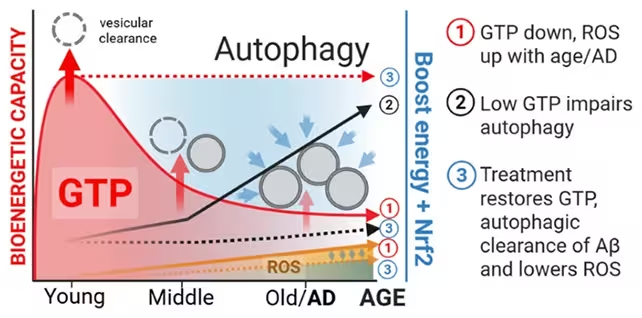
The study emphasizes that the observed effects were strongest when EGCG and nicotinamide were applied together, suggesting synergy: EGCG’s antioxidant and signaling effects may complement nicotinamide’s role in nucleotide and NAD+ metabolism, jointly supporting GTP restoration.
Implications for Alzheimer’s research and treatment development
The work highlights GTP as an underappreciated driver of neuronal housekeeping functions. If GTP depletion contributes to the slow accumulation of misfolded proteins in aging brains, therapies that restore nucleotide balance could slow or prevent downstream pathology, including amyloid aggregation linked to Alzheimer’s.
While EGCG and nicotinamide are accessible through diet and supplements, the authors and other experts caution that these results arise from in vitro mouse-cell models. Translating such findings to humans requires careful dose optimization, pharmacokinetic studies, and controlled clinical trials to determine safety and efficacy for preventing or modifying neurodegenerative disease.
Earlier epidemiological work has connected regular green tea consumption to fewer white matter lesions and lower dementia risk, although causality there remains unproven. Nicotinamide and related NAD+ precursors have shown neuroprotective effects in stroke models and other neurodegeneration studies. This new mechanistic link to GTP provides a plausible biochemical explanation for some of those observations and offers a concrete biochemical target for further research.
Expert Insight
Dr. Amina Patel, Professor of Neurobiology at the Institute for Brain Health (fictional), comments: "The UC Irvine findings are compelling because they shift attention to nucleotide energy states—GTP specifically—as a modifiable factor in neuronal maintenance. We need to see whether similar GTP deficits occur in human neurons in aging and Alzheimer’s brains, and whether dietary or supplement approaches can achieve therapeutic concentrations in the human central nervous system."
Dr. Patel adds: "It’s encouraging that the compounds are already characterized for safety in many contexts, but clinical translation will require rigorous trials. This study gives us a clear biochemical hypothesis to test."
Limitations, safety and next steps
Key limitations to keep in mind:
- The experiments were conducted in cultured mouse neurons, not in living animals or humans. Cellular responses can differ markedly in intact brain tissue.
- Effective concentrations in vitro may not be achievable or safe in humans without formulation changes or delivery strategies that improve brain bioavailability.
- Long-term effects, interactions with other medications, and potential off-target consequences of chronically manipulating nucleotide pools are unknown.
Planned next steps include validating the GTP response in animal models of aging and Alzheimer’s, elucidating the precise biochemical pathways by which EGCG and nicotinamide increase GTP, and designing translational studies that address dosing, blood-brain barrier penetration and safety in older adults.
Related technologies and research directions
- Nutraceutical formulations and prodrugs that enhance CNS delivery of EGCG or nicotinamide derivatives.
- Biomarkers to monitor neuronal GTP levels or autophagic activity in vivo.
- Combination strategies that pair metabolic support with anti-amyloid or anti-tau therapeutics.
Conclusion
UC Irvine’s study identifies a promising biochemical route—restoring neuronal GTP via a green tea polyphenol (EGCG) plus nicotinamide—that reactivates autophagy and reduces amyloid beta accumulation in cultured mouse neurons. The results illuminate a potential mechanism linking diet, supplementable metabolites and brain proteostasis, and they support further preclinical and clinical research. While encouraging, these findings remain preliminary: controlled animal studies and human trials will be required to determine whether this approach can translate into safe, effective prevention or treatment strategies for Alzheimer’s disease.
Source: sciencealert


Leave a Comment