6 Minutes
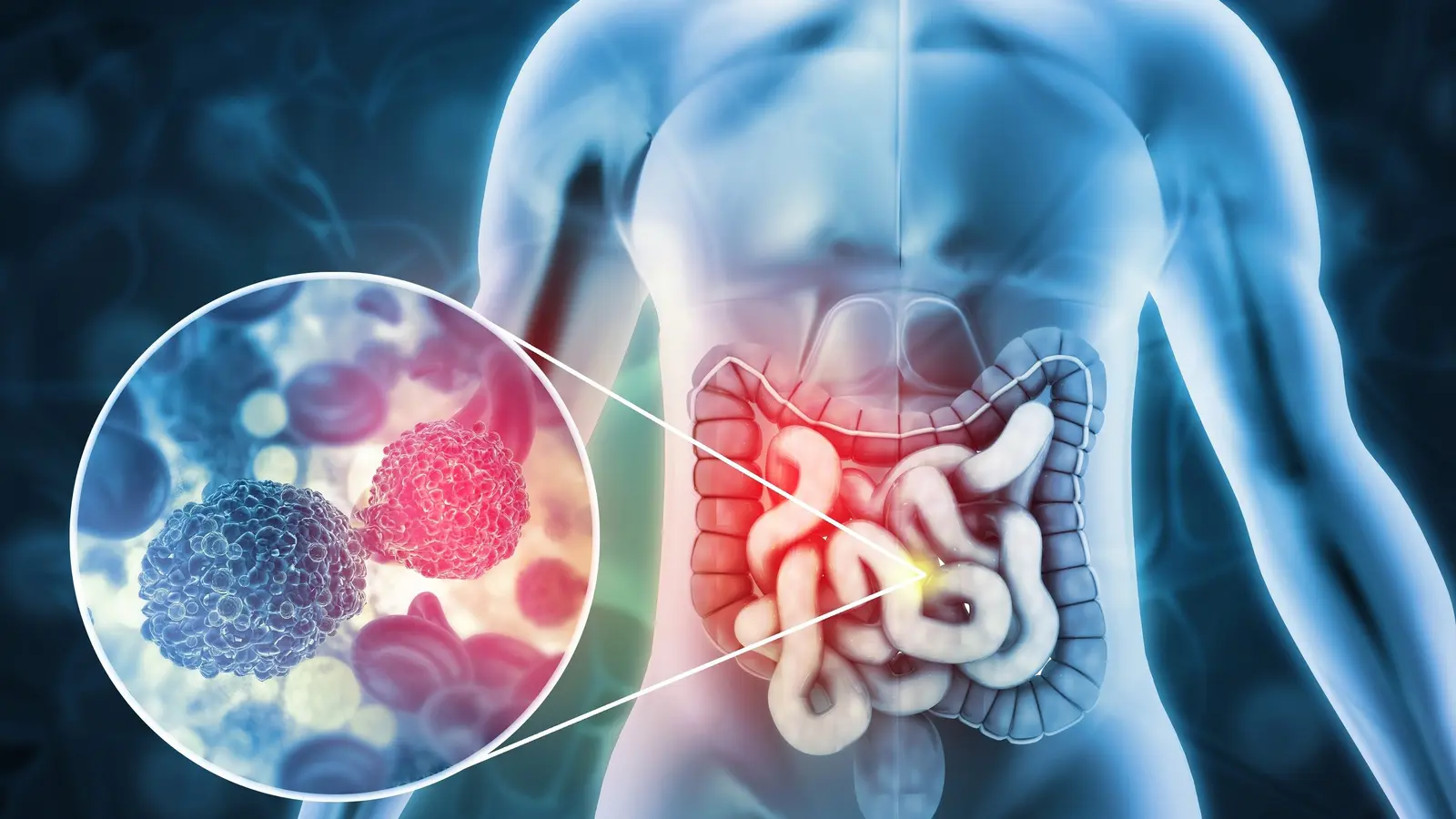
Microbes left a signature where scientists least expected a clear pattern: inside the DNA data of tumours. The idea is simple, almost cunning—when hospitals sequence cancer genomes, they intend to read human DNA, but those same files also carry tiny fragments of viral and bacterial DNA. A recent reanalysis of thousands of whole-genome sequences shows that one cancer type, colorectal cancer, consistently carries a microbial fingerprint strong enough to set it apart from other tumours.
It feels a bit like eavesdropping on the microscopic passengers that hitch a ride with tumor samples. In a dataset that pooled sequencing from projects including Genomics England, researchers sifted through more than 11,700 cancer samples across 22 cancer types, and a pattern emerged: colorectal tumours repeatedly harbored distinct microbial communities. Other cancers, in contrast, did not show the same reproducible signal.
How the signal was teased out
Sequencing labs are noisy. Human DNA drowns out microbial sequences by orders of magnitude, and lab contamination can mimic genuine biology. So the challenge is not merely looking; it is separating the whispers from the shout. The team developed computational filters and quality controls that strip away human reads, flag common contaminants, and assemble a reliable profile of which microbes are truly present in each sample.
Data and method highlights
The analysis combined whole genome sequencing (WGS) files from thousands of patients, drawing on public and clinical repositories. Rather than adding a separate swab or infection panel, the approach mines what’s already in the sequencing output. That makes it efficient: as WGS becomes routine in clinical oncology, microbial readouts could be obtained at little extra cost. The researchers cross-referenced microbial profiles with clinical metadata—tumour type, staging and outcomes—to look for patterns that might matter to diagnosis or prognosis.
Why might the colon show a consistent microbial signature while other organs do not? The colon is among the most microbe-rich habitats in the human body. It hosts a dense, diverse microbiome that interacts intimately with the mucosal lining. That ecological richness appears to leave a stronger, more reproducible trace in tumor tissue than cancers that arise in relatively sterile environments.
Key discoveries and clinical implications
Perhaps the clearest finding: colorectal tumours exhibit a microbial community profile that can distinguish them from other cancer types with high accuracy. Think of it as a biological watermark—subtle, but distinctive. Clinicians sometimes face a diagnostic puzzle when metastatic disease is discovered but the primary tumour is unknown; a microbial fingerprint might help point pathologists toward a colorectal origin in ambiguous cases.
Beyond origin tracing, the sequencing approach flagged clinically relevant viral infections in other cancers. For example, human papillomavirus (HPV) was reliably detected in oral cancer samples, matching the results one would expect from dedicated clinical tests. The incidental detection of viruses such as HTLV-1—rare but clinically meaningful—also emerged from the data. These are the kinds of findings that can influence treatment pathways or prompt additional screening for associated conditions.
There were hints of prognostic value as well. In certain sarcoma cases, the presence or absence of specific bacteria correlated with patient survival. In some instances particular bacteria associated with poorer outcomes, while in others they aligned with better survival. Those relationships are intriguing. They do not yet prove causation, but they open avenues for targeted research: Could microbes modulate immune responses to therapy? Could they serve as biomarkers that refine prognosis or guide individualized treatments?
Professor Daniel Brewer of Norwich Medical School highlighted the clinical upside: whole genome sequencing is maturing into more than a way to read human mutations. It can also detect hidden infections and provide context that influences diagnosis and patient care. As sequencing becomes embedded in routine oncology workflows, extracting microbial information is a low-friction add-on with potentially high value.
Broader scientific context
This work also reframes a popular idea in the field—that each cancer type carries its own unique microbiological signature. The new analysis suggests that this is not universally true. Some cancers may lack a consistent microbial community, or the signal may be too weak or heterogeneous to reproduce across cohorts. The colon, because of its microbiome ecology, appears to be an exception rather than the rule.
That distinction matters. It tempers broad claims about a universal tumour microbiome and pushes the conversation toward nuance: which cancers reliably carry microbial information, under what conditions, and how should clinicians and researchers validate such signals before clinical use?
Expert Insight
"The demonstration that one cancer type—colorectal—retains a reproducible microbial imprint in standard sequencing data is a practical breakthrough," says Dr. Maya Jennings, a microbial ecologist at a major cancer research centre. "It shows that genomic medicine can borrow valuable ecological data for diagnostic and prognostic ends. But caution is needed: technical rigor and independent validation are essential before this becomes part of routine decision-making."
There are technological and scientific next steps. Larger, geographically diverse cohorts must be tested to ensure the fingerprint holds across populations. Labs need standardized pipelines to distinguish signal from contamination. And mechanistic studies are required to move from association to understanding: are microbes passengers, or active participants in tumour biology?
For patients, the immediate promise is practical. Many cancer patients already undergo WGS as part of precision oncology. If microbial analysis is layered onto that existing workflow, clinicians could gain insights into tumour origin, infectious co-factors and potentially prognosis—without collecting new samples. The strategy turns one dataset into two, squeezing more value from the same clinical resource.
Science often advances by listening to what was previously dismissed as background noise. In cancer genomics, the microbes hiding in sequencing files have gone from nuisance to possible allies. The next phase will determine whether that microbial whisper becomes a routine part of how we diagnose and treat cancer.
Source: scitechdaily
Comments
skyspin
is this even true? sounds like contamination risk or overfitting. curious if it holds across countries and labs, idk tho, needs more proof.
bioNix
wow, microbes hiding in cancer genome files? wild. if this helps find primaries faster that's huge, but lab contamination worries me. need more validation and diverse cohorts.

Leave a Comment