8 Minutes
Introduction: The hidden power of scent
Smell is one of the least obvious senses, yet it exerts outsized influence over memory, emotion and behaviour. In culture and cinema, a single aroma can unlock a flood of recollections; in biology, olfaction guides feeding, danger avoidance and social interaction. But when smell fades or becomes distorted, the change may reflect more than a nuisance — it can be an early indicator of neurodegenerative disease.
This article examines the science behind olfactory dysfunction (anosmia and hyposmia), why smell loss can precede diseases like Parkinson's, and how clinicians and researchers are exploring scent as a practical biomarker for earlier diagnosis and better patient care.
How smell works: a short scientific primer
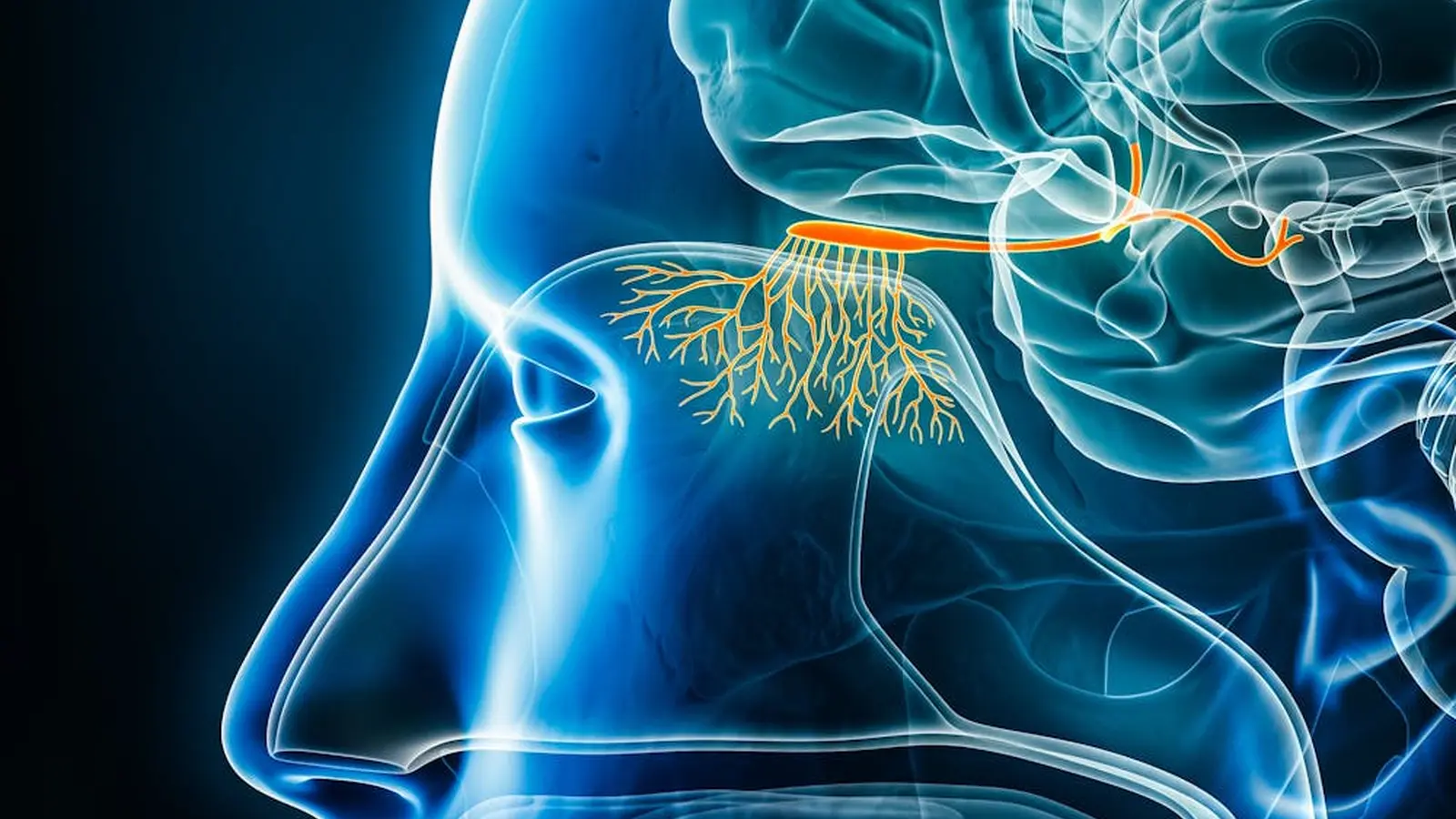
Olfaction begins at the nasal epithelium, where receptor neurons bind volatile molecules and transmit signals into the brain. Those signals first reach the olfactory bulb, a compact forebrain structure closely linked to regions that manage memory and emotion — notably the limbic system and hippocampus. Because of these direct connections, smells are uniquely capable of evoking vivid memories.
When an odorant binds to receptors, sensory neurons send patterns of activity to the olfactory bulb, which encodes and relays that information to higher cortical areas for identification and emotional tagging. Damage anywhere along this pathway — from the receptor neurons to central processing centers — can alter detection, discrimination and perception of odors.
Common causes of smell loss and distortion
Smell loss is frequently temporary and benign: viral infections (such as common colds or influenza), nasal polyps, allergic inflammation and head trauma are typical causes. During the COVID-19 pandemic, anosmia and parosmia (distorted smells) brought widespread attention to olfactory disorders when many patients reported transient loss followed by months of altered perception.
Not all olfactory dysfunction is peripheral. Central nervous system changes — including neurodegenerative processes — can produce smell loss long before more recognizable symptoms such as motor impairment or cognitive decline appear.
Olfactory loss as an early sign of Parkinson's disease
Parkinson's disease (PD) is a progressive neurodegenerative disorder best known for motor symptoms: tremor, rigidity and slowness of movement. By the time these signs emerge, substantial loss of dopamine-producing neurons in the substantia nigra has often already occurred. That delay makes early detection difficult.
Olfactory dysfunction is common in Parkinson's: studies indicate up to 80–90% of patients exhibit measurable smell impairment, often years before motor symptoms manifest. The leading hypothesis is that misfolded proteins and pathological changes begin in peripheral neural structures — possibly the olfactory bulb and related brainstem nuclei — and later propagate to motor areas. Environmental exposures such as inhaled pesticides, airborne toxins or viral agents have been proposed as initiators of damage to the olfactory system, although mechanisms vary across individuals and remain under investigation.
Crucially, smell loss is not specific to Parkinson's; it also appears in Alzheimer's disease, normal ageing and numerous ENT conditions. That limits its diagnostic specificity as a lone marker but does not eliminate its value: combined with other clinical data and biomarkers, olfactory testing can improve early detection and risk stratification.
Patterns of olfactory change in Parkinson's
Not all odors are affected equally in Parkinson's disease. Research shows a selective deficit: patients often struggle to detect neutral or unpleasant smells (e.g., smoke, rubber, certain cleaning agents) while some pleasant aromas (e.g., chocolate) are comparatively preserved. Some individuals also experience phantosmia — phantom smells that are not present — or parosmia, where familiar odors are distorted and unpleasant.
Anecdotal reports and controlled studies suggest Parkinson's may even have a characteristic odor signature, described by some as woody or musky. One well-known case involved a person with exceptional olfactory sensitivity who detected an unusual scent years before her spouse's clinical diagnosis; subsequent research has sought to quantify and validate such scent-based signals.
Clinical applications: testing and diagnostic value
Clinicians can use standardized smell tests (scratch-and-sniff or odor identification batteries) to quantify olfactory function. When combined with imaging, genetic screening and other biomarkers, olfactory testing can help differentiate Parkinson's from atypical parkinsonian syndromes and predict disease progression. For example, pronounced smell loss has been associated with a higher likelihood of classic Parkinson's pathology and with greater nonmotor burden.
However, clinicians must interpret olfactory results in context. Age-related decline, smoking history, chronic sinus disease and medications all modify smell perception. A measured, multimodal diagnostic approach improves specificity: olfactory testing can provide an early alert that prompts longitudinal monitoring, neuroimaging, or referral to specialized care.
Research directions and future prospects
Researchers are pursuing several promising avenues:
- Biomarker integration: Combining olfactory testing with molecular biomarkers (alpha-synuclein assays, neurofilament light chain), neuroimaging and digital motor assessments to build predictive models for Parkinson's.
- Mechanistic studies: Tracing how pathological proteins may spread from olfactory structures to deeper brain regions to clarify causal pathways and identify intervention points.
- Therapeutic timing: Using smell loss as an early warning to enroll at-risk individuals in neuroprotective trials before major neuronal loss occurs.
- Olfactory rehabilitation: Developing interventions to restore or retrain smell perception after viral injury or neurodegeneration, improving safety and quality of life.
Several longitudinal cohort studies are tracking large groups of individuals with olfactory impairment to determine which patterns predict conversion to Parkinson's or other disorders. These efforts aim to translate olfactory science into actionable clinical tools.
Expert Insight
"Olfactory dysfunction gives us a rare, accessible window into early brain change," says Dr. Elena Morales, neurologist and professor of neurodegenerative disease research at the Institute for Brain Health. "It isn't a standalone diagnosis, but when we see persistent, unexplained smell loss—especially alongside subtle REM sleep changes or constipation—it raises our index of suspicion. Early identification allows monitoring and, potentially, earlier therapeutic intervention when disease-modifying treatments become available."
Implications for patients and caregivers
Loss or distortion of smell affects safety (reduced ability to detect smoke or gas), nutrition (reduced appetite and enjoyment of food) and mental health (depression and social withdrawal). Patients experiencing persistent, unexplained changes in smell should discuss the symptom with primary care or neurology. Simple smell tests and a careful history can identify those who need follow-up imaging or referral to a movement disorders specialist.
Public awareness is also relevant: distinguishing transient post-viral anosmia from progressive olfactory decline matters for timely evaluation. Education campaigns and accessible screening tools could help at-risk individuals seek earlier assessment.
Conclusion
Smell loss is common, multifactorial and often underappreciated. In the context of Parkinson's disease and other neurodegenerative disorders, olfactory dysfunction frequently appears years before hallmark symptoms, offering a potential early-warning sign. While smell testing alone cannot confirm Parkinson's, it is a low-cost, noninvasive component of a layered diagnostic strategy that could improve early detection, patient monitoring and ultimately clinical outcomes as disease-modifying therapies emerge.
Ongoing research aims to convert olfactory observations into robust, clinically actionable biomarkers. For patients, caregivers and clinicians, paying attention to changes in scent perception is a practical step toward earlier recognition and better management of neurodegenerative risk.
Source: sciencealert

Leave a Comment