4 Minutes
Study overview and key image
A new study from the Florey Institute of Neuroscience and Mental Health in Melbourne reports that COVID-19 infection in male mice produced measurable changes in sperm that were associated with increased anxiety-like behaviours in their offspring. The peer-reviewed findings, published in Nature Communications, indicate molecular alterations in sperm RNA and persistent changes in brain gene activity that could reflect epigenetic inheritance.
"We found that the resulting offspring showed more anxious behaviours compared to offspring from uninfected fathers," said Elizabeth Kleeman, the study's first author. Co-senior author Carolina Gubert noted that altered gene activity in the hippocampus — a brain region central to emotion regulation — may help explain the behavioural changes.
Experimental design and molecular findings
Researchers exposed adult male mice to the virus that causes COVID-19, then mated those males with uninfected females and tracked the health, behaviour, and brain gene expression of the resulting litters. All offspring sired by infected fathers displayed increased anxiety-like behaviours in standard behavioural tests, the team reported.
The study identified modifications to specific RNA molecules in sperm after infection. These RNA changes included species known to influence gene networks involved in neural development. According to the authors, some of these sperm-borne RNA molecules are implicated in regulating genes active during early brain formation.
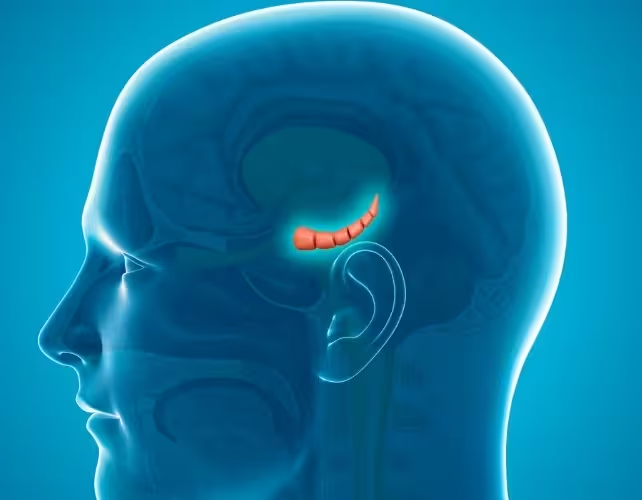
The hippocampus (centre) in a human brain
In female offspring, the team observed "significant changes" in hippocampal gene expression patterns. Carolina Gubert summarized the interpretation: "This may contribute to the increased anxiety we observed in offspring, via epigenetic inheritance and altered brain development." Lead researcher Anthony Hannan emphasized the broader possible implications: "These findings suggest that the COVID-19 pandemic could have long-lasting effects on future generations." He also cautioned that more work is needed to determine whether the same mechanisms operate in humans.
Scientific context and implications
Epigenetic inheritance refers to non-DNA-sequence changes — such as chemical marks on DNA or RNA content in germ cells — that can influence gene expression in descendants. Environmental factors including infection, stress, and diet have been shown in animal models to change sperm epigenetic marks and offspring traits. This study adds SARS-CoV-2 infection to that list in a controlled mouse model.
If similar processes occur in people, the public-health implications could be substantial: millions of individuals worldwide were infected during the pandemic, and transgenerational effects would extend the pandemic's biological footprint beyond those directly ill. However, the authors and outside experts stress caution. Human germline and developmental biology differ from mice, and epidemiological evidence linking parental COVID-19 to altered neurodevelopment in children is not yet established.
Next steps and research priorities
Future research should test whether comparable sperm RNA and epigenetic changes appear in infected human males, whether these signals persist over time, and how they interact with maternal factors and postnatal environment. Longitudinal cohort studies and mechanistic lab work will be needed to translate these animal findings into clinical recommendations.
Conclusion
The Melbourne study provides experimental evidence in mice that paternal SARS-CoV-2 infection can modify sperm molecular content and coincide with anxiety-related changes in offspring brain function and behaviour. While the results raise important questions about possible transgenerational effects of infectious disease, confirming relevance to humans will require careful, multidisciplinary follow-up.
Source: sciencealert
Comments
Tomas
Feels overhyped tbh. Solid mouse work but anxiety assays can be finicky, and translating to people is a big leap. still, follow up needed
atomwave
wow didn't expect that... kinda scary to think infections could ripple into future generations. hope they check humans next, pronto!
labcore
Hmm, interesting but is this even true for humans? mice sperm RNA changes sound plausible, yet human germline is different, and mothers + environment matter too.

Leave a Comment