5 Minutes
New target for bone strengthening identified
A multidisciplinary team from the University of Leipzig (Germany) and Shandong University (China) has identified a cell-surface receptor, GPR133 (also known as ADGRD1), as a critical regulator of bone formation. The discovery, reported in Signal Transduction and Targeted Therapy, shows that activating this receptor stimulates osteoblasts—the cells responsible for building bone—and can substantially increase bone mass and mechanical strength in mouse models.
Genetic variants in GPR133 had been previously associated with differences in bone mineral density in human genetic studies, prompting researchers to investigate the receptor’s biological role. Using a combination of gene-deletion models and a small-molecule activator called AP503, the team tested how loss or stimulation of GPR133 affected skeletal development and bone maintenance.
Experimental approach and key findings
The study used mice engineered either to lack GPR133 or to retain a version of the receptor that could be pharmacologically stimulated. Animals missing the receptor developed reduced bone mass and structural fragility resembling osteoporotic changes. By contrast, mice given AP503—which was identified via a computer-assisted screen as a GPR133 agonist—showed increased osteoblast activity, greater bone formation, and measurable improvements in bone strength.
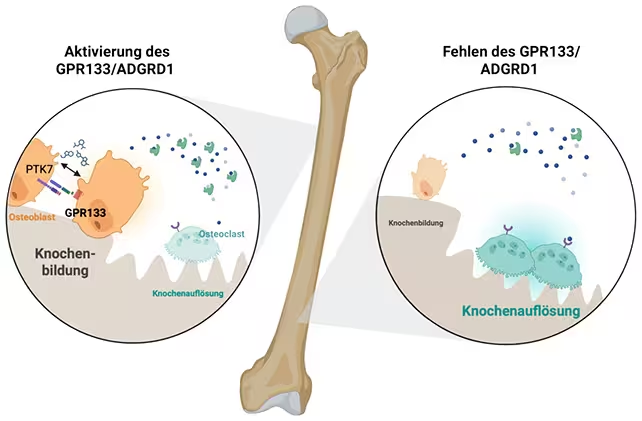
With GPR133 activated (on the left), the osteoblasts (orange) are more dominant. (Biorender, Ines Liebscher)
AP503 behaved like a molecular "on" switch for osteoblasts, increasing bone deposition in both healthy mice and models with bone loss. The researchers also tested combinations of receptor activation with mechanical loading (exercise-like stimuli) and observed additive effects: GPR133 stimulation plus physical activity produced greater gains in bone mass and resilience than either intervention alone.
Mechanism in brief
GPR133 is a member of the adhesion G protein–coupled receptor family. These receptors transduce signals across the cell membrane to trigger intracellular pathways that control cell differentiation and function. In this study, activating GPR133 enhanced osteoblast differentiation and activity, shifting the balance toward bone formation over resorption. Although the detailed downstream signaling cascade requires further mapping, the result is an increased rate of new bone matrix deposition and improved microarchitecture.
Implications for osteoporosis and aging
Osteoporosis is a chronic condition that weakens bone and raises fracture risk; it affects millions worldwide and is particularly common after menopause. Current therapies can slow further bone loss but generally cannot restore degraded bone to its prior state without risks or diminishing efficacy over time. Identification of GPR133 as a druggable receptor opens a potential route for anabolic treatments that rebuild bone rather than only reducing bone resorption.
The Leipzig team emphasizes that results to date derive from animal models, but the conserved nature of osteoblast biology suggests translational potential. If GPR133 function is compromised by genetic variation or age-associated decline, receptor-targeted therapies could be developed to enhance bone density in at-risk populations, including postmenopausal women and older adults.
"By pharmacologically stimulating GPR133, we observed notable increases in bone strength across models," said Ines Liebscher, a biochemist involved in the research. Molecular biologist Juliane Lehmann noted the receptor’s promise for medical applications targeting skeletal fragility in aging populations.
Related technologies and next steps
Translating this discovery into clinical treatments will require several steps: optimization of GPR133 agonists for potency and safety, detailed toxicology, and rigorous preclinical studies in larger animals before human trials. Researchers will also need to determine long-term effects, dosing strategies, and whether activation can safely reverse established osteoporosis without off-target consequences.
Expert Insight Dr. Maria Andersen, an endocrinologist and bone biology researcher unaffiliated with the study, commented: "This work offers an exciting anabolic strategy. If GPR133 agonists can be refined and shown safe in humans, they could complement exercise and nutrition interventions to rebuild bone in patients who currently have limited options. The pathway-specific approach reduces the likelihood of broad systemic effects, but careful clinical evaluation is essential."
Conclusion
The identification of GPR133 as a regulator of osteoblast-driven bone formation provides a promising new avenue for osteoporosis research. In mice, activating the receptor with a small molecule (AP503) increased bone formation and strength and worked synergistically with physical loading. While translation to human therapy will require extensive further testing, targeting GPR133 could eventually yield anabolic treatments that restore degraded bone and reduce fracture risk in aging populations.
Source: sciencealert

Leave a Comment